| 22:23 UK time, Monday, 28 September 2009
 Researchers in Oxford have started the first trial of Britain's two swine flu vaccines. Working with teams in Bristol, Exeter, Southampton and London, they are aiming to recruit 1,000 youngsters aged six months to 12 years. Although both vaccines are likely to get licensed in the next few weeks, none of the trials has taken place in Britain. Pandemrix (made by GSK) was approved by the European Medicines Agency on Friday, while Baxter's H1N1 jab seems likely to get approved this week.
Researchers in Oxford have started the first trial of Britain's two swine flu vaccines. Working with teams in Bristol, Exeter, Southampton and London, they are aiming to recruit 1,000 youngsters aged six months to 12 years. Although both vaccines are likely to get licensed in the next few weeks, none of the trials has taken place in Britain. Pandemrix (made by GSK) was approved by the European Medicines Agency on Friday, while Baxter's H1N1 jab seems likely to get approved this week.This is the first time the vaccines have been used in the UK and the first comparative study of the jabs.
 Professor Andrew Pollard from the University of Oxford is heading the research: "We are doing this trial to compare head-to-head the two vaccines which are going to be used in the UK against swine flu to see which one works best in children and which one is best tolerated. We are sort of in a race against time because we know the flu season is already started. We hope to immunise the children over the next 10 days or so.
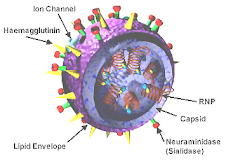
Professor Andrew Pollard from the University of Oxford is heading the research: "We are doing this trial to compare head-to-head the two vaccines which are going to be used in the UK against swine flu to see which one works best in children and which one is best tolerated. We are sort of in a race against time because we know the flu season is already started. We hope to immunise the children over the next 10 days or so.The GSK vaccine is made using the traditional means of growing the virus in embryonated hens' eggs, before the virus is broken and deactivated. The Baxter vaccine is made in cell culture, so is suitable for those who have an egg allergy. The sort of side effects that are expected are sore arms and fevers. The information given to parents also lists rare side effects including Guillian-Barré syndrome which can cause ascending paralysis and even be fatal.
It's worth quoting here from the trial information: "Other very rare events that have been seen with routine flu vaccines include seizures and temporary bleeding disorders. In the past Guillian-Barré syndrome (a rare disorder of nerves) has been associated with flu vaccines but the relationship remains uncertain, with some studies suggesting a possible link but others not finding it. One large study in the UK found that influenza-like illness itself was associated with an increased risk of the Guillian-Barré syndrome but there was no link with the seasonal influenza vaccines, suggesting that vaccination might actually protect against the disorder by preventing flu."
What I take that to mean is that, if you have the vaccine, there is a remote, theoretical possibility that you could get GBS (as happened in the USA in 1976 with their swine flu vaccine), but you are more likely to get it as a result of contracting flu. So while it is impossible to rule out the risk of GBS from the vaccine, it is very remote, and this must be balanced against the very real and proven risks of complications from flu.